Newsletter
22 de December de 2023
2024-2025 Regulatory Agenda Has Been Approved by the Brazilian FDA
ANVISA’s new Regulatory Agenda 2024-2025 (RA 24/25) was approved and published on the Official Gazette on December 18th. The Joint Ordinance No. 1.409 of 2023 approved the Regulatory Agenda (RA) which covers the priorities for the 2024-2025 biennium.
The Regulatory Agenda is a planning instrument for the normative activities of the Agency. It presents several priority topics to be regulated by ANVISA whilst its validity. Therefore, such Agenda sheds light on the themes do to be improved by regulatory means for the next two years in Brazil.
Social participation alongside the National Sanitary Surveillance System (Sistema Nacional de Vigilância Sanitária – SNVS) was crucial to the construction of the RA 24/25. There were 1.449 contributions received by ANVISA’s Public Consultancy.
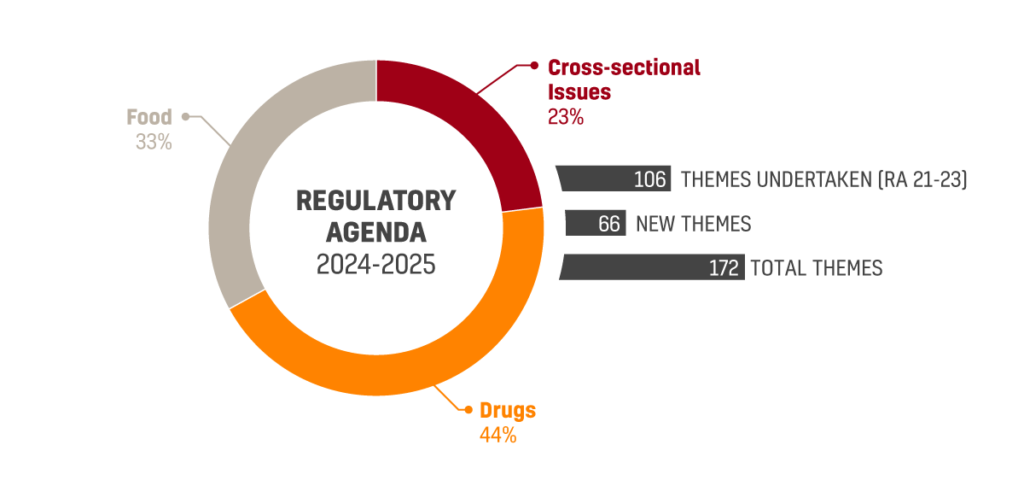
The just approved agenda covers 172 regulatory themes, divided into 16 macro themes. Topics that received most regulatory demands include Drugs with 45 themes, Food with 34 themes and cross-sectional issues with 23 themes.
106 out of the 172 themes pertaining the new agenda are related to the subject matters already announced on the 2021-2023 agenda. 66 themes are new and unrelated to the previous agenda.
The Regulatory Agenda 2024-2025 guarantees visibility and transparency for the normative regulatory activities by ANVISA. It will come into effect on January 2nd, 2024.
For more information regarding ANVISA, please do not hesitate to contact us by email: regulatorio@kasznarleonardos.com.
Last related news
15 de May de 2026
CONAR updates Influencers Guide in line with the new requirements for transparency and protection in the digital environment
The National Council for Advertising Self-Regulation (“CONAR”) released, last Monday (11), the new edition of the Guide to Marketing and Advertising by … CONAR updates Influencers Guide in line with the new requirements for transparency and protection in the digital environment

11 de May de 2026
Digital ECA: Signs of Practical Enforcement of the New Regulatory Framework
The Digital Statute of the Child and Adolescent (Law No. 15,211/2025 – “Digital ECA”) came into effect in March 2026 and is … Digital ECA: Signs of Practical Enforcement of the New Regulatory Framework

27 de March de 2026
New Structure of the ANPD and Impacts Brought by the Digital Child and Adolescent Statute
Among the most recent publications related to the Brazilian Data Protection Agency (“ANPD”), the release of Decree No. 12.881/2026 stands out. The Decree approves … New Structure of the ANPD and Impacts Brought by the Digital Child and Adolescent Statute




